In this first blog in a series of 3, we introduce our latest report on the impact of COVID-19 on patient access to cancer care in Europe in which we map the negative impact and identify lessons learned. More details about the content of the report will follow in our next blogs. Stay tuned!
In 2020, EFPIA published Every Day Counts, an overview of 10 systemic factors which are delaying patient access to innovative oncology therapies in Europe, and the implications of those delays on patients’ lives. That report, which you can access here, was the result of a multi-stakeholder collaboration between 2019 and early 2020. It established a collective understanding of the causes of delays in patient access.
When the report was published, European countries were past the first peak of the coronavirus pandemic and restriction measures were being eased. All the stakeholders involved in the development of the report had experienced the impact of this novel disease at first hand, and the effects of the resulting pandemic on patient access to cancer care.
With the second wave underway in late 2020, many of the stakeholder organizations proposed an addendum which could take a high-level view of the impact of the pandemic on patient access to oncology therapies in Europe.
The report addendum was compiled in partnership with more than 19 European organizations and references more than 90 documents.
Although our stakeholders had experienced the negative effects of the pandemic for themselves, it was nevertheless a shock to see the full picture of these aggregated effects and to appreciate the sheer scale of the impact COVID-19 has had on cancer care.
A detrimental impact on patient access to cancer care across europe
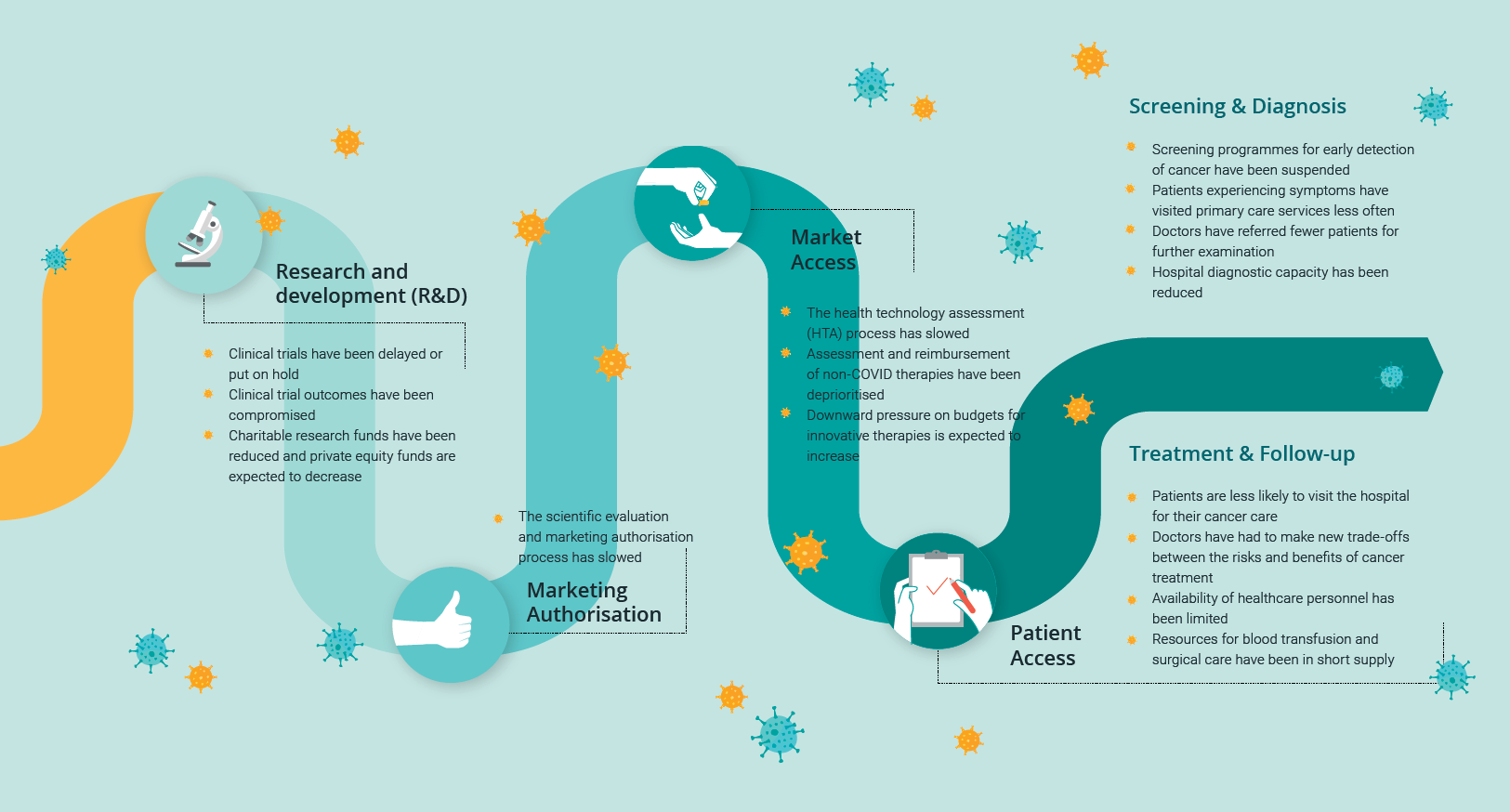
The report addendum provides an overview of the ways in which COVID-19 has had a negative impact on patient access to oncology services in Europe. The disruptions have affected the status of all four key milestones identified in the original report as necessary to ensure access to innovative oncology therapies:
- Research & Development
- Marketing Authorisation
- Market Access
- Patient Access
The total of 15 disruptions ranges from the suspension of screening programs to the increased pressure on doctors to weigh up the benefits and potential risks of treating oncology patients in hospital environments where COVID-19 exposure is a serious risk. We will dive deeper into the 15 identified effects in our next blog.

Projections suggest a severe impact of COVID-19 on cancer mortality
The exact impact on cancer deaths has yet to become clear, but projections suggest it may be severe.
- A global study found that a treatment delay of four weeks is associated with a 6-13% increase in the risk of death. Delays of up to twelve weeks further increase this risk. In breast cancer, for example, an eight-week delay in surgery increases the risk of death by 17%, while a twelve-week delay increases it by 26% (Hanna et al., 2020).
- Seven comprehensive care centers in Europe reported that in April 2020 the number of cancer patients admitted fell by 20-30% (Van de Haar et al, 2020).
- Surgery was the most affected modality being delayed or cancelled in more than 10% of patients in 34% of the centers, whereas early cessation of palliative treatment was reported in 32.1% of the centers – Findings from a survey among 109 medical oncologists from 18 countries in Europe (Onesti et al., 2021)
Never let a crisis go to waste
Though the negative effects were substantial and will likely be felt for years to come, the pandemic has also been an environment which has generated key learnings. This information could be used to make cancer care delivery both more resilient to disruptions caused by a crisis of similar magnitude, and more sustainable in a post-pandemic world.
“In the normal setting, the last step of the Marketing Authorization process (EMA opinion to European Commission decision) has a formal duration of 67 days. During the pandemic, this step was taken within a few hours.” – Member of the EOP European Multi-Stakeholder Sounding Board
Our report addendum provides six key recommendations as to how European healthcare systems can recover from the impact of the COVID-19 crisis and adapt to both minimise access delays for current patients and reduce the impact of future crises on the cancer patients of tomorrow:
- Clear the cancer backlog now, using innovative practices which emerged during the pandemic
- Maintain the proven agility of R&D and Marketing Authorization processes
- Continue the intensified European collaboration in clinical assessment to use scarce HTA resources more efficiently after the pandemic
- Continue the adoption of digital health to increase remote care and use healthcare resources more efficiently after the pandemic
- Maintain and build adaptive surge capacity to be ready for future disruptions to cancer care
- Safeguard cancer budgets as a critical enabler for improving continuity, efficiency, and sustainability of cancer care.
We will dive deeper into these recommendations in our follow-up blog in this series of blogs.
“Instead of having a patient come in so we can dispense an investigational drug in a vial, we now ship it to them overnight.” – Oncologist (Lee McFarling, 2020)
What have we Learned?
With cancer increasing as a global primary cause of death and source of increase in health expenditure, we have the opportunity to learn from the experience of this healthcare crisis, capture this common spirit and sense of urgency and unify efforts to recover from the COVID-19 pandemic as soon as possible and make cancer care delivery more resilient to future disruptions and sustainability challenges.
In capturing and examining learnings from investigators, government agencies, pharmaceutical companies, professional societies, and patient organisations about the impact of COVID-19 on patient access to cancer care in Europe, we have discovered both the threats posed by the pandemic’s impact on oncology care and the positive developments and learnings which have arisen because of efforts to mitigate these detrimental effects.
“The fact a disease can be managed collaboratively, rationally, and systematically is something we have never experienced.” – Member of the EOP European Multi-Stakeholder Sounding Board
let’s discuss
Inspired to share your thoughts? Or would you like to learn more on how to implement the learnings from the pandemic? We would be delighted to hear from you. Please feel invited to contact Bas Amesz.
DO YOU WANT TO READ MORE?
Please also be invited to read the articles in our “Every day counts” blog series, which we published previously:
- White paper release: Every day counts
- Blog: Bringing stakeholders together to improve patient access to oncology therapies in Europe
- Blog: European access discussions shouldn’t stop at reimbursement
- Blog: Deep dive: The Patient Access Indicator
- Blog: The 10 key factors delaying patient access across Europe
- Blog: Introducing new cancer treatments: how reducing time to Market Access dramatically impacts patients’ lives
- Blog: Speeding up patient access requires dealing with uncertainty on clinical value first, negotiating price second



